Health
Women’s Underwear Found to Contain Harmful Chemicals in New Lab Tests
A recent investigation by consumer watchdog groups from Hungary, Austria, Slovenia, and the Czech Republic has revealed dangerously high levels of endocrine-disrupting chemicals in women’s underwear. The study, which included contributions from the Czech environmental advocacy group Arnika, focused on bisphenols—synthetic chemicals often found in plastics that have been linked to serious health risks.
Bisphenols, particularly bisphenol A (BPA), are widely used in industrial manufacturing, including food packaging, medical devices, and textiles. BPA is classified as a hazardous chemical by the European Union and has been linked to a range of health issues, including fertility problems, hormonal imbalances, and cognitive impairments. The watchdog groups tested 166 types of women’s underwear from Hungary, Austria, and Slovenia for the presence of these chemicals, with concerning results.
The study found that 30% of the tested underwear samples contained bisphenols, while 10% had levels higher than what is considered safe for human health by regulatory bodies. Interestingly, the tests revealed that bisphenol levels were higher in underwear from well-known brands compared to cheaper alternatives. However, cotton underwear was found to be largely free from bisphenols, regardless of the brand.
“Although cotton products are available for women, our initial market research showed that the majority of women’s panties are made from synthetic materials,” said Júlia Dénes, a chemist with the Hungarian Association of Conscious Consumers. This reliance on synthetic fabrics may increase the risk of exposure to harmful chemicals, according to the researchers.
The health risks associated with bisphenol A are well-documented. The European Environment Agency has warned that the chemical can negatively impact fertility, cause allergic reactions, damage eyesight, disrupt hormonal systems, and affect cognitive function and metabolism. While most exposure to BPA comes from food and drink packaging, experts believe that it can also be absorbed through the skin—raising concerns about its presence in everyday clothing items.
This study adds to growing evidence of widespread bisphenol contamination. Last year, EU-funded research found bisphenol A in the urine of 92% of people tested across 11 European countries, with many cases exceeding European safety limits. While BPA is already banned in certain products, such as baby bottles and food packaging for young children, it remains prevalent in many consumer goods.
In June, EU member states supported a plan to ban bisphenol A in food and drink packaging across the bloc. However, environmental advocates argue that the regulatory framework is lagging behind scientific evidence. “Manufacturers are replacing BPA with other bisphenols, which pose similar health risks and are not yet banned,” said Karolina Brabcova, consumer campaigns manager with Arnika’s toxics and waste programme.
Advocates are calling for stricter regulations, urging authorities to ban bisphenols and other harmful chemicals from all consumer products to better protect public health.
Health
Europe Struggles to Meet Tuberculosis Targets Amid Rising Drug Resistance
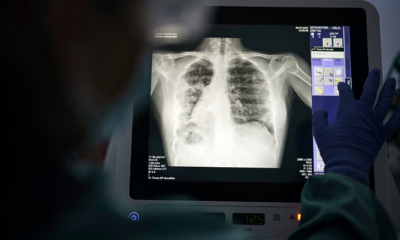
Tuberculosis remains a public health issue in Europe as the region falls behind elimination targets, according to a new report by the World Health Organization (WHO) and the European Centre for Disease Prevention and Control (ECDC). The report warns that one in five cases goes undiagnosed, while drug resistance continues to exceed global averages.
Although the overall number of TB cases has declined, progress toward elimination remains insufficient. Tuberculosis, an infectious disease spread when an individual with pulmonary TB expels bacteria through coughing, is the leading cause of death from a single infectious agent worldwide. Common symptoms include a persistent cough lasting more than two weeks, fever, night sweats, and weight loss.
Across the WHO European Region, which covers 53 countries in Europe and Central Asia, TB incidence has fallen by 39 percent since 2015, and deaths have dropped by 49 percent. These figures fall short of the WHO’s End TB Strategy 2025 milestones of 50 percent reduction in cases and 75 percent reduction in deaths.
Within the European Union, cases have decreased by 33 percent and deaths by 17 percent, insufficient to meet the 2030 targets. Health authorities note this gap has led to thousands of preventable infections and fatalities. In 2024, more than 160,000 newly diagnosed TB cases were reported, while the estimated actual number of cases was 204,000, meaning only 79 percent were officially notified.
Hans Kluge, WHO Regional Director for Europe, said undiagnosed cases represent missed opportunities to treat patients earlier and curb transmission. “By investing in rapid diagnosis, shorter all-oral treatment regimens and stronger follow-up, countries can reach more people earlier, improve outcomes and put us back on track toward our targets,” he said. Delayed diagnosis increases treatment difficulty and heightens the risk of spreading the disease.
Drug-resistant tuberculosis remains a major challenge. Multi-drug resistant TB among new and previously treated cases stands at 23 percent and 51 percent, respectively, far above the global averages of 3.2 percent and 16 percent. Standard treatment for non-resistant TB typically lasts six months with success rates above 85 percent, but resistant variants require longer, more complex regimens with lower success rates.
Most European countries now report low TB incidence, with notification rates below 10 per 100,000. The disease mainly affects vulnerable populations, including migrants, prison inmates, and people with HIV co-infections. Ralf Otto-Knapp of the German Central Committee against Tuberculosis warned that lower overall case numbers in Western Europe have led to decreased attention, making it harder to combat multidrug-resistant TB. “We must strengthen treatment and prevention services, ensure easy access to new medicines and foster cross-border cooperation,” he said.
The report underscores that without urgent action on diagnosis, treatment, and drug resistance, Europe risks falling further behind its TB elimination goals, putting vulnerable communities at heightened risk.
Health
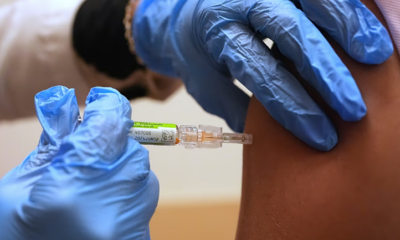
Kent Meningitis B Outbreak Cases Drop to 29, UKHSA Confirms
The number of confirmed cases in the Kent meningitis B outbreak has fallen to 29 following laboratory reclassification, according to United Kingdom health authorities. This marks a decrease from the 34 cases reported on Saturday, according to the latest update from the UK Health Security Agency (UKHSA).
The number of confirmed deaths remains at two. One victim was a 21-year-old student at the University of Kent, and the other a sixth-form pupil from nearby Faversham.
“Some cases initially classified as confirmed have been reclassified following further laboratory results and clinical investigation,” the agency said on Sunday. The UKHSA expects additional probable cases may also be downgraded in the coming days as further laboratory assessments are completed.
Sherine Thomas, infectious diseases consultant at UKHSA, said authorities are maintaining close monitoring and working with NHS England and local authorities to respond rapidly to any new cases. “We continue to remain vigilant for new cases and work closely with NHS England and local authorities across the country, to ensure that any new cases identified are responded to as quickly as possible,” Thomas said.
The outbreak has raised concerns in Kent, particularly among families and educational institutions. Health authorities have reiterated that children born before May 1, 2015, are not eligible for the MenB vaccine unless they fall into a clinical risk group. Parents and guardians have been urged to seek medical advice if they notice symptoms such as fever, headache, nausea, or neck stiffness.
The European Centre for Disease Prevention and Control (ECDC) has assessed the risk to the general population in the European Union and European Economic Area (EU/EEA) as very low, citing the small probability of exposure and infection. Public health officials in the UK continue to emphasize that while the outbreak remains serious locally, the threat to the broader population is minimal.
Local authorities in Kent have also increased awareness campaigns and targeted vaccination programs in affected areas, focusing on schools and universities. Healthcare providers have been advised to be vigilant for early symptoms and to provide timely treatment to prevent severe outcomes.
The UKHSA stressed that maintaining close monitoring, rapid laboratory testing, and vaccination of at-risk groups remain central to controlling the outbreak. Families in the region have expressed relief at the reduction in confirmed cases but remain cautious as investigations continue.
The agency continues to advise the public that early detection and medical attention are crucial in managing meningitis B, which can progress rapidly and lead to serious complications if untreated.
Health
Airbrush Tanning Solution Sun Labs for Smooth, Even Bronze Color

Airbrush tanning solution sun labs is a popular choice for people who want a rich bronze finish without sun exposure. It delivers fast color, dries quickly, and works well for both home users and salon professionals. Because the formula develops evenly, many users choose it when they want reliable tanning results.
The product is often selected for special events, vacations, and weekly beauty routines. In addition, it blends well on many skin tones. That flexibility makes it useful for beginners who want simple application and professionals who need dependable color.
Why Airbrush Tanning Solution Sun Labs Is Popular
One reason many people prefer this formula is its fast visible effect. The cosmetic bronzer gives immediate color, while the active tanning ingredient continues developing for several hours. As a result, users can see coverage quickly and still gain deeper color later.
Another benefit is how smoothly the mist applies. Since the liquid is designed for spray systems, it reaches skin evenly when used correctly. Therefore, streaks are easier to avoid compared with heavy creams.
Main Benefits at a Glance
-
Quick drying texture
-
Even spray distribution
-
Deep bronze tone
-
Easy layering for darker color
-
Suitable for face and body
-
Works in many airbrush systems
Many users also appreciate that the formula feels light after application. It does not leave a thick coating, so skin remains comfortable during development.
How to Prepare Skin Before Spraying
Preparation improves final tanning results. First, exfoliate dry areas carefully because rough skin absorbs more color. Knees, elbows, ankles, and hands need extra attention before any spray session.
Next, avoid lotions, oils, or heavy creams before applying the tanning solution. These products may block even absorption. Instead, clean dry skin creates the most balanced finish.
A simple preparation routine often includes:
-
Shower several hours before tanning
-
Shave at least one day earlier if possible
-
Wear loose dark clothing
-
Remove deodorant and perfume
-
Keep skin completely dry
Because preparation affects color quality, skipping these steps can shorten wear time.
Best Application Methods for Even Coverage
When spraying airbrush tanning solution sun labs, distance matters. Hold the spray gun several inches from the body and move continuously. This creates a fine layer instead of heavy wet spots.
Start with lighter passes. Then allow each section to settle before adding more solution. In this way, color builds gradually and remains natural.
For home users, this order often works best:
-
Legs
-
Arms
-
Torso
-
Back
-
Face
Face application should stay lighter because facial skin often develops color faster.
Areas That Need Less Product
Some body parts absorb tanning solution more quickly. Therefore, use less spray on:
-
Hands
-
Feet
-
Elbows
-
Knees
-
Neck folds
A soft blending mitt can help soften edges after spraying.
Development Time and Color Results
After application, color continues developing for several hours. Most people leave the solution on skin between six and eight hours before rinsing lightly.
During that period, avoid sweating or moisture because water can interrupt development. Therefore, staying indoors helps protect even color.
Expected color depth often depends on skin tone:
-
Light skin: soft golden bronze
-
Medium skin: warm tan finish
-
Deeper skin: enhanced bronze depth
Some users apply a second layer later for stronger darkness.
How Long the Tan Usually Lasts
A well-prepared application can remain visible for five to seven days. However, daily habits strongly affect longevity.
For example, hot showers and harsh scrubs remove color faster. Gentle skincare extends the appearance much longer.
Ways to Extend Wear Time
-
Use mild body wash
-
Pat skin dry after bathing
-
Apply lightweight moisturizer daily
-
Avoid exfoliating too soon
-
Limit chlorinated swimming
Because hydrated skin sheds more evenly, the tan fades in a smoother way.
Choosing the Right Shade Strength
Some users prefer one coat for natural warmth, while others want darker results. Fortunately, spray layering allows both options.
A lighter session often suits daytime wear or first-time users. Meanwhile, deeper passes create stronger evening color.
When selecting intensity, consider:
-
Natural skin tone
-
Event timing
-
Clothing color
-
Desired contrast
Testing a small area first helps predict final development.
Common Mistakes to Avoid
Heavy spraying in one area often causes uneven darkness. Therefore, steady movement is more important than speed.
Another common mistake is dressing too quickly. Tight fabrics may rub fresh bronzer before it settles.
Avoid these errors:
-
Spraying too close
-
Applying on damp skin
-
Skipping exfoliation
-
Using too much on dry areas
-
Showering too early
Even experienced users benefit from consistent technique.
Why Many Users Prefer Sun Labs Formulas
Many tanning users return to this formula because color appears strong without looking orange when applied correctly. That balance is important for both daily wear and formal events.
In addition, the formula adapts well to machine spraying. Professional technicians often value predictable mist flow and stable consistency.
Features Frequently Mentioned by Users
-
Smooth mist texture
-
Reliable dark bronze tone
-
Quick visible guide color
-
Easy layering control
These qualities help explain why it remains a repeat purchase.
Aftercare for Better Fade Quality
After the first rinse, skin should be treated gently. The first wash removes cosmetic bronzer while developed color remains.
Moisturizing later in the day helps preserve the finish. As skin naturally renews, hydration slows uneven fading.
A simple aftercare routine includes:
-
First rinse without soap
-
Wait before heavy exercise
-
Moisturize morning and night
-
Avoid exfoliation for several days
Because aftercare controls fade quality, it matters almost as much as application.
Final Thoughts on Airbrush Tanning Solution Sun Labs
Airbrush tanning solution sun labs offers a practical way to create rich bronze color with controlled application. It suits both new users and experienced spray tanning routines.
With proper skin preparation, even spraying, and careful aftercare, results often look smoother and last longer. For anyone seeking dependable spray tanning performance, this formula remains a strong option for regular use.
-

 Entertainment2 years ago
Entertainment2 years agoMeta Acquires Tilda Swinton VR Doc ‘Impulse: Playing With Reality’
-

 Business2 years ago
Business2 years agoSaudi Arabia’s Model for Sustainable Aviation Practices
-

 Business2 years ago
Business2 years agoRecent Developments in Small Business Taxes
-

 Home Improvement1 year ago
Home Improvement1 year agoEffective Drain Cleaning: A Key to a Healthy Plumbing System
-

 Politics2 years ago
Politics2 years agoWho was Ebrahim Raisi and his status in Iranian Politics?
-

 Business2 years ago
Business2 years agoCarrectly: Revolutionizing Car Care in Chicago
-

 Sports2 years ago
Sports2 years agoKeely Hodgkinson Wins Britain’s First Athletics Gold at Paris Olympics in 800m
-

 Business2 years ago
Business2 years agoSaudi Arabia: Foreign Direct Investment Rises by 5.6% in Q1