Health
Study Finds US Doctors Prescribe ADHD Drugs Too Quickly for Young Children
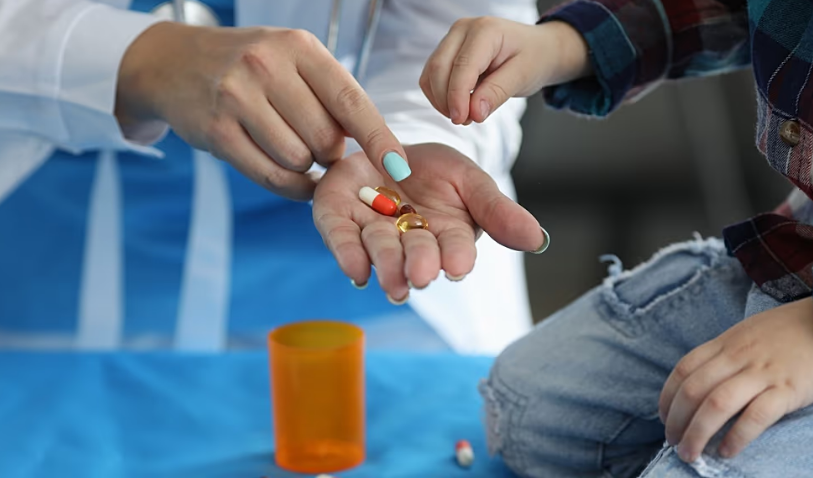
A new study has found that many young children in the United States diagnosed with attention-deficit/hyperactivity disorder (ADHD) are being prescribed medication almost immediately, despite medical guidelines recommending behavioural therapy as the first step.
The analysis, published in JAMA Network Open, reviewed the cases of more than 9,700 children and revealed that 42.2% of four- and five-year-olds received ADHD medication within one month of diagnosis. By contrast, only 14.1% began drug treatment more than six months after diagnosis, the timeframe recommended by health experts.
Current US medical guidelines state that children aged four and five should undergo at least six months of behavioural therapy before starting on stimulants such as Ritalin or Concerta. These therapies aim to build skills and habits to help children manage daily challenges. Medication is considered a next step if behavioural treatment alone is not effective.
“This practice is concerning, because we know starting ADHD treatment with a behavioural approach is beneficial,” said Dr Yair Bannett, lead author of the study and assistant professor of paediatrics at Stanford University. “It has a big positive effect on the child as well as on the family.”
ADHD is one of the most common developmental disorders in children, affecting around 11.4% of Americans aged three to 17. Symptoms typically begin before age 12 and may include hyperactivity, restlessness, difficulty concentrating, and forgetfulness. Left untreated, ADHD can significantly affect academic performance and long-term outcomes into adulthood.
Experts note that while a combination of behavioural therapy and medication is often the most effective treatment, young children in particular should first be given non-drug interventions. However, access to behavioural therapy remains a challenge in many parts of the country, which may contribute to doctors prescribing medication sooner than recommended.
The study emphasised that safety concerns were not the primary issue. While ADHD medications are generally considered safe for young children, many families discontinue use due to side effects such as irritability, aggression, or heightened emotional reactions. “We never think of medication as the only solution for ADHD,” Bannett said, stressing that behavioural support is an essential component of treatment.
The researchers also highlighted limitations of the study, noting that it focused solely on the United States. ADHD prescribing patterns can vary significantly by region, with previous research suggesting children in North America are more likely to receive stimulant medications compared with their peers in Europe.
As the debate continues, the findings add weight to calls for expanding access to behavioural therapy, ensuring young children diagnosed with ADHD receive treatment in line with medical guidelines.
Health
Global Mental Health Cases Near 1.2 Billion as Anxiety and Depression Drive Sharp Worldwide Rise

A major global analysis has found that mental health conditions have surged to an estimated 1.2 billion people worldwide, driven largely by steep increases in anxiety and depression over the past three decades.
The findings, published in The Lancet as part of the Global Burden of Disease Study 2023, show that the number of people living with mental disorders has almost doubled since 1990, marking a 95% rise. Researchers say major depressive disorder and anxiety disorders have seen even sharper growth, increasing by 131% and 158% respectively, making them the most prevalent mental health conditions globally.
The report describes mental illnesses as widespread conditions that create long-term disability and significant human suffering. It also highlights broader consequences for economies and public services, including reduced productivity, lower workforce participation and increasing pressure on health and welfare systems.
Researchers estimate that in 2023 alone, around 620 million females and 552 million males were affected by mental health conditions. While the overall burden is rising across both sexes, the study points to notable differences in the types and prevalence of disorders.
Among women, depression and anxiety were the most commonly reported conditions, alongside higher rates of eating disorders such as anorexia nervosa and bulimia nervosa. The report links this disparity to a mix of biological, social and structural factors, including exposure to domestic violence, sexual abuse, gender inequality and reproductive health-related changes.
In contrast, neurodevelopmental and behavioural disorders, including attention deficit hyperactivity disorder (ADHD), conduct disorder and autism spectrum conditions, were more frequently diagnosed in men.
Teenagers aged 15 to 19 were identified as the group experiencing the highest mental health burden globally, raising concerns about early onset of conditions and insufficient preventive care for young people.
The study identifies several key risk factors associated with mental illness, including childhood sexual violence, bullying and intimate partner violence. These factors are strongly linked to conditions such as depression, schizophrenia, bipolar disorder and anxiety disorders. However, researchers note that such exposures have remained relatively stable over time and account for only a portion of the overall rise.
According to the authors, broader drivers are likely contributing to the increasing prevalence of mental disorders. These include genetic and biological influences, poverty, inequality, and the growing impact of global crises such as armed conflict, pandemics, natural disasters and climate-related stress.
While mental health conditions have long been a leading cause of disability worldwide, the report warns that the situation is worsening. At the same time, health systems have not expanded services at a pace matching demand.
The authors caution that the gap between rising need and limited access to care is becoming more pronounced, leaving millions without adequate treatment or support.
Health
Europe Records Sharp Rise in Sexually Transmitted Infections, ECDC Warns
Health
White House Rejects Report It Blocked Return of Ebola-Infected US Doctor
-

 Entertainment2 years ago
Entertainment2 years agoMeta Acquires Tilda Swinton VR Doc ‘Impulse: Playing With Reality’
-

 Sports2 years ago
Sports2 years agoChina’s Historic Olympic Victory Sparks National Pride Amid Controversy
-

 Business2 years ago
Business2 years agoSaudi Arabia’s Model for Sustainable Aviation Practices
-

 Business2 years ago
Business2 years agoRecent Developments in Small Business Taxes
-

 Home Improvement1 year ago
Home Improvement1 year agoEffective Drain Cleaning: A Key to a Healthy Plumbing System
-

 Politics2 years ago
Politics2 years agoWho was Ebrahim Raisi and his status in Iranian Politics?
-

 Sports2 years ago
Sports2 years agoKeely Hodgkinson Wins Britain’s First Athletics Gold at Paris Olympics in 800m
-

 Business2 years ago
Business2 years agoCarrectly: Revolutionizing Car Care in Chicago