Health
Counterfeit COVID-19 and Influenza Test Kits Reach European Market
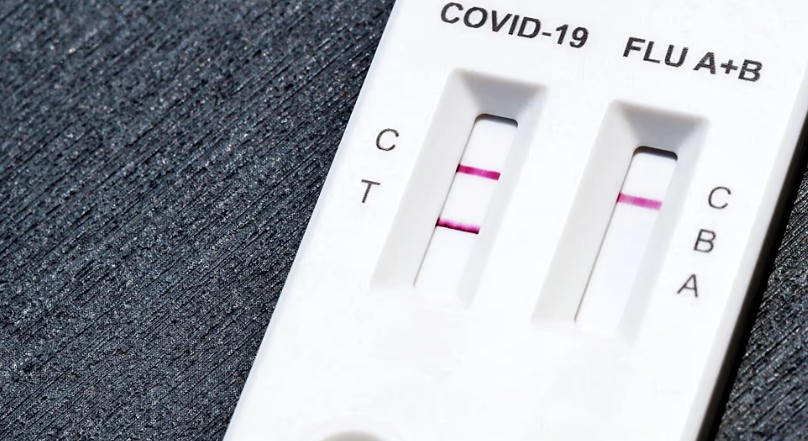
Manipulated units of a combined test for COVID-19 and influenza have reached the European market. The manufacturer has confirmed that batch FCO24090516 is fake, raising concerns about the safety and reliability of these self-testing kits.
The Spanish Agency for Medicines and Health Products (AEMPS) was alerted by Portuguese authorities about the circulation of counterfeit tests that claim to detect both coronavirus and influenza A+B. The legitimate product is manufactured by Safecare Biotech, based in Hangzhou, China. However, the detected units bear the batch number FCO24090516, which Safecare Biotech has confirmed as counterfeit.
While the AEMPS has launched an investigation to trace the distribution of these manipulated units in Spain, authorities have said there is no evidence so far that they have reached pharmacies or official distributors in the country. The presence of legitimate tests with the same reference number, FCO-6032, makes it challenging to distinguish fake units at a glance.
Authorities have highlighted three key indicators for identifying counterfeit kits. First, the label shows altered information, listing batch FCO24090516, a manufacture date of September 2024, and an expiry date of September 2026. Second, the test cassette and the tube containing the extracting solution lack the lot number and dates that appear on authentic units. Third, the swab included with the kit differs from the one supplied by Dalian Rongbang Medical, the manufacturer represented in Europe by Lotus NL.
Health officials stress that these products should only be purchased from pharmacies to ensure their authenticity. Buying kits from unofficial sources carries risks related to origin, storage conditions, and lack of professional guidance. Anyone who already possesses one of these suspect tests is advised not to use it.
Experts have warned that the adulteration of medical devices is a serious concern. These devices play a critical role in personal and public health, guiding decisions about isolation, treatment, and seeking medical care. Using a counterfeit test could produce incorrect results, potentially leaving infections undetected or leading to unnecessary treatments.
The AEMPS is coordinating with European authorities to monitor the situation and prevent further distribution of counterfeit kits. The agency also reminds the public that even minor alterations in test kits can compromise their effectiveness, reinforcing the need for vigilance and caution when purchasing medical products.
As investigations continue, health authorities emphasize that self-testing remains a valuable tool in managing COVID-19 and influenza, but only when the kits are authentic and used according to instructions. Counterfeit products not only undermine public trust in medical testing but also pose real risks to individual and collective health.
-

 Entertainment2 years ago
Entertainment2 years agoMeta Acquires Tilda Swinton VR Doc ‘Impulse: Playing With Reality’
-

 Business2 years ago
Business2 years agoSaudi Arabia’s Model for Sustainable Aviation Practices
-

 Business2 years ago
Business2 years agoRecent Developments in Small Business Taxes
-

 Sports2 years ago
Sports2 years agoChina’s Historic Olympic Victory Sparks National Pride Amid Controversy
-

 Home Improvement1 year ago
Home Improvement1 year agoEffective Drain Cleaning: A Key to a Healthy Plumbing System
-

 Politics2 years ago
Politics2 years agoWho was Ebrahim Raisi and his status in Iranian Politics?
-

 Sports2 years ago
Sports2 years agoKeely Hodgkinson Wins Britain’s First Athletics Gold at Paris Olympics in 800m
-

 Business2 years ago
Business2 years agoCarrectly: Revolutionizing Car Care in Chicago