Health
Toxic ‘Forever Chemicals’ Found in Reusable Menstrual Products, Raising Health Concerns
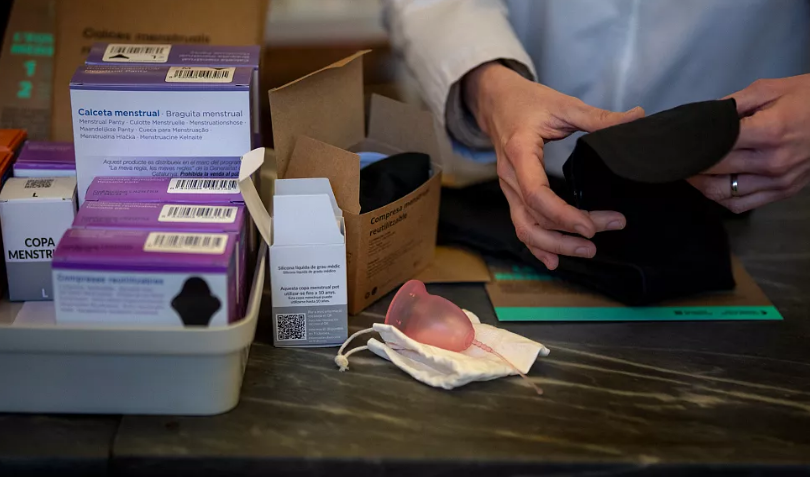
A new scientific study has found evidence of potentially harmful “forever chemicals” in reusable menstrual and incontinence products, raising alarms about the safety of widely used items marketed as eco-friendly alternatives.
Published in the journal Environmental Science and Technology Letters, the peer-reviewed study examined 59 products from North America, South America, and Europe. Researchers found that nearly 30% contained elevated levels of fluorine, a marker used to identify per- and polyfluoroalkyl substances (PFAS)—a group of synthetic chemicals known for their durability and resistance to degradation.
Period underwear and reusable pads were found to contain the highest concentrations of PFAS, suggesting they were added intentionally during manufacturing to provide properties like water resistance. In contrast, other items like menstrual cups and incontinence products showed lower levels, indicating possible incidental contamination.
PFAS, often dubbed “forever chemicals,” are associated with a range of health risks, including immune suppression, liver damage, fertility issues, and certain cancers. Two PFAS compounds—PFOA and PFOS—are classified by the World Health Organization as carcinogenic or possibly carcinogenic, and are banned in the European Union.
“These chemicals are not necessary to make menstrual products functional,” said Marta Venier, one of the study’s authors and an environmental chemist at Indiana University. “We identified several products with no intentional PFAS, showing it is possible to manufacture these items without using harmful substances.”
Health experts warn that the presence of PFAS in menstrual products is especially concerning due to the prolonged and intimate skin contact involved. “Skin is a good barrier, but in the case of menstrual products, we are dealing with the vaginal area, which is more permeable,” explained Tina Kold Jensen, a physician and environmental epidemiologist at the University of Southern Denmark.
Jensen noted that exposure to PFAS through hygiene products could lead to long-term accumulation in the body, posing a risk to reproductive health and even affecting newborns through placental transfer or breast milk.
While reusable menstrual products are generally promoted for their environmental benefits, this study underscores the need for stricter regulation and clearer labeling of chemical content. According to a 2022 survey in Spain, reusable menstrual products were more popular than disposable options among women aged 26 to 35.
Government initiatives, like Catalonia’s free menstrual product scheme, aim to reduce waste and period poverty—but researchers stress the importance of ensuring such products are free of toxic substances.
The issue of PFAS in menstrual products is not new. In 2023, UK-based consumer group Which? found high silver levels in some popular period underwear brands. In the U.S., Thinx faced a class-action lawsuit after PFAS were detected in its products. The case was settled in 2022.
Health
Global Mental Health Cases Near 1.2 Billion as Anxiety and Depression Drive Sharp Worldwide Rise

A major global analysis has found that mental health conditions have surged to an estimated 1.2 billion people worldwide, driven largely by steep increases in anxiety and depression over the past three decades.
The findings, published in The Lancet as part of the Global Burden of Disease Study 2023, show that the number of people living with mental disorders has almost doubled since 1990, marking a 95% rise. Researchers say major depressive disorder and anxiety disorders have seen even sharper growth, increasing by 131% and 158% respectively, making them the most prevalent mental health conditions globally.
The report describes mental illnesses as widespread conditions that create long-term disability and significant human suffering. It also highlights broader consequences for economies and public services, including reduced productivity, lower workforce participation and increasing pressure on health and welfare systems.
Researchers estimate that in 2023 alone, around 620 million females and 552 million males were affected by mental health conditions. While the overall burden is rising across both sexes, the study points to notable differences in the types and prevalence of disorders.
Among women, depression and anxiety were the most commonly reported conditions, alongside higher rates of eating disorders such as anorexia nervosa and bulimia nervosa. The report links this disparity to a mix of biological, social and structural factors, including exposure to domestic violence, sexual abuse, gender inequality and reproductive health-related changes.
In contrast, neurodevelopmental and behavioural disorders, including attention deficit hyperactivity disorder (ADHD), conduct disorder and autism spectrum conditions, were more frequently diagnosed in men.
Teenagers aged 15 to 19 were identified as the group experiencing the highest mental health burden globally, raising concerns about early onset of conditions and insufficient preventive care for young people.
The study identifies several key risk factors associated with mental illness, including childhood sexual violence, bullying and intimate partner violence. These factors are strongly linked to conditions such as depression, schizophrenia, bipolar disorder and anxiety disorders. However, researchers note that such exposures have remained relatively stable over time and account for only a portion of the overall rise.
According to the authors, broader drivers are likely contributing to the increasing prevalence of mental disorders. These include genetic and biological influences, poverty, inequality, and the growing impact of global crises such as armed conflict, pandemics, natural disasters and climate-related stress.
While mental health conditions have long been a leading cause of disability worldwide, the report warns that the situation is worsening. At the same time, health systems have not expanded services at a pace matching demand.
The authors caution that the gap between rising need and limited access to care is becoming more pronounced, leaving millions without adequate treatment or support.
Health
Europe Records Sharp Rise in Sexually Transmitted Infections, ECDC Warns
Health
White House Rejects Report It Blocked Return of Ebola-Infected US Doctor
-

 Entertainment2 years ago
Entertainment2 years agoMeta Acquires Tilda Swinton VR Doc ‘Impulse: Playing With Reality’
-

 Sports2 years ago
Sports2 years agoChina’s Historic Olympic Victory Sparks National Pride Amid Controversy
-

 Business2 years ago
Business2 years agoSaudi Arabia’s Model for Sustainable Aviation Practices
-

 Business2 years ago
Business2 years agoRecent Developments in Small Business Taxes
-

 Home Improvement1 year ago
Home Improvement1 year agoEffective Drain Cleaning: A Key to a Healthy Plumbing System
-

 Politics2 years ago
Politics2 years agoWho was Ebrahim Raisi and his status in Iranian Politics?
-

 Sports2 years ago
Sports2 years agoKeely Hodgkinson Wins Britain’s First Athletics Gold at Paris Olympics in 800m
-

 Business2 years ago
Business2 years agoCarrectly: Revolutionizing Car Care in Chicago